In continuation of my update on tofacitinib
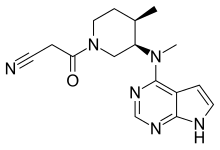
Pfizer Inc. announced that the United States Food and Drug Administration (FDA) has approved Xeljanz 5 mg twice daily (BID) and Xeljanz XR (tofacitinib) extended release 11 mg once daily (QD) for the treatment of adult patients with active psoriatic arthritis (PsA) who have had an inadequate response or intolerance to methotrexate or other disease-modifying antirheumatic drugs (DMARDs). Xeljanz/Xeljanz XR is the first and only Janus kinase (JAK) inhibitor approved by the FDA for both moderate to severe rheumatoid arthritis (RA) and active PsA.
“Psoriatic arthritis is a complex and progressive disease with an unpredictable course,” said Angela Hwang, Global President, Inflammation and Immunology, Pfizer. “The approval of Xeljanz is an important step forward for patients seeking new treatments and is a testament to Pfizer’s unwavering commitment to advancing patient care.”
The recommended dose of Xeljanz/Xeljanz XR is in combination with nonbiologic DMARDs, and use in combination with biologic DMARDs or with potent immunosuppressants such as azathioprine and cyclosporine is not recommended.
The FDA approval of Xeljanz for the treatment of adult patients with active PsA was based on data from the Phase 3 Oral Psoriatic Arthritis Trial (OPAL) clinical development program, which consisted of two pivotal studies, OPAL Broaden and OPAL Beyond, as well as available data from an ongoing long-term extension trial, OPAL Balance. The findings from OPAL Broaden and OPAL Beyond were published in October 2017 in the New England Journal of Medicine.
Both pivotal studies met their two primary efficacy endpoints, demonstrating statistically significant improvements in American College of Rheumatology 20 (ACR20) response and change from baseline in the Health Assessment Questionnaire–Disability Index (HAQ-DI) score at three months in patients receiving Xeljanz 5 mg BID treatment in combination with a nonbiologic DMARD, compared to those treated with placebo. In OPAL Broaden, 50% of patients taking Xeljanz 5 mg BID achieved an ACR20 response, compared to 33% of patients taking placebo (p≤0.05), at three months. In OPAL Beyond, 50% of patients achieved an ACR20 response with Xeljanz 5 mg BID, compared to 24% of patients taking placebo (p≤0.05), at three months. In both studies, statistically significant improvements in ACR20 response was also seen with Xeljanz 5 mg BID compared to placebo at week 2, a secondary endpoint and the first post-baseline assessment (OPAL Broaden: 22% and 6% [p=0.0003], respectively; OPAL Beyond: 27% and 13% [p=0.0046], respectively).
“As a practicing rheumatologist, I’ve seen the significant physical impact psoriatic arthritis has on people living with the disease, and many patients are looking for additional therapeutic options,” said Philip Mease, M.D., Swedish Medical Center, University of Washington and study investigator. “I’m pleased that Xeljanz is now available for use in the treatment of this chronic condition.”
The safety profile observed in patients with active psoriatic arthritis treated with Xeljanz was consistent with the safety profile observed in rheumatoid arthritis patients. The most common adverse events observed occurring in greater than 3% of patients on Xeljanz 5 mg BID were nasopharyngitis, upper respiratory tract infection, headache and diarrhea.
“Psoriatic arthritis is a serious and debilitating chronic illness that should be diagnosed and treated early,” said Randy Beranek, president and CEO, National Psoriasis Foundation. “As an organization that advocates for people living with psoriatic arthritis, we welcome the availability of new therapies for treating this disease.”