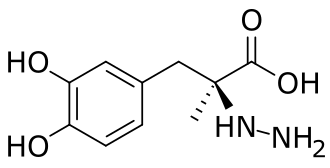
PTC Therapeutics, Inc. (NASDAQ: PTCT) announced U.S. Food and Drug Administration (FDA) approval of Sephience™ (sepiapterin) for the treatment of children and adults living with phenylketonuria (PKU). The approval includes broad labeling for the treatment of hyperphenylalaninemia (HPA) in adult and pediatric patients 1 month of age and older with sepiapterin-responsive PKU.
"We are excited to have reached this important milestone for those affected by PKU," said Matthew B. Klein, M.D., Chief Executive Officer of PTC Therapeutics. "The broad labeling reflects the potential of Sephience to meet the significant unmet need of PKU patients. The Sephience clinical data along with our expertise in launching rare disease therapies position Sephience to become the future standard of care. Our experienced customer facing teams are ready to bring this therapy to children and adults with PKU in the United States as quickly as possible."
The FDA approval is based on the evidence of significant efficacy and safety from the
Phase 3 APHENITY trial as well as durability of treatment effect in the APHENITY long-term extension study.
"The approval marks an exciting milestone for the PKU community," said Catherine Warren, Executive Director of the National PKU Alliance. "This progress brings renewed hope, and we are eager to see the positive impact this new treatment option will have on advancing care and potentially improving quality of life for individuals of all ages and PKU subtypes that respond to this therapy."
Sephience was recently granted marketing authorization by the European Commission. Review of approval applications is ongoing in several other countries including Japan and Brazil.
About Sephience (sepiapterin)
Sephience is indicated for the treatment of hyperphenylalaninemia (HPA) in adult and pediatric patients with phenylketonuria (PKU). Sephience is a natural precursor of the enzymatic co-factor BH4, a critical co-factor for phenylalanine hydroxylase (PAH). Through its mechanism of action, Sephience is able to effectively reduce blood phenylalanine (Phe) levels and has the potential to treat a broad range of PKU patients. Sephience is approved in the European Economic Area and the United States.
Indication and Important Safety Information
Indication
Sephience is indicated for the treatment of hyperphenylalaninemia (HPA) in adult and pediatric patients 1 month of age and older with sepiapterin-responsive phenylketonuria (PKU). Sephience is to be used in conjunction with a phenylalanine (Phe)-restricted diet.
Contraindications
None
Important Safety Information
Treatment with Sephience should be directed by physicians knowledgeable in the management of PKU. Biochemical response to Sephience can only be determined by a therapeutic trial with careful monitoring of ongoing dietary and nutritional balance to ensure adequate Phe control.
Warnings and Precautions
Increased Bleeding: Sephience may increase the risk of bleeding. Bleeding events, including superficial hematomas, prolonged bleeding, and heavy menstrual bleeding have occurred in patients treated with Sephience. Inform patients about the risk of bleeding associated with Sephience and have patients follow up with their healthcare provider should such a bleeding event occur. Consider treatment interruption with Sephience in patients with active bleeding.
Hypophenylalaninemia: Some pediatric patients receiving Sephience experienced hypophenylalaninemia. Monitor blood Phe levels during treatment and modify the dosage of Sephience and/or dietary protein and Phe intake as needed to ensure adequate blood Phe level control. Frequent blood monitoring is recommended in the pediatric population.
Interaction with Levodopa: In a 10-year post-marketing safety surveillance program for a non-PKU indication using another drug that is a phenylalanine hydroxylase (PAH) activator, 3 patients with underlying neurological disorders experienced seizures, exacerbation of seizures, over-stimulation, and irritability during co-administration with levodopa. Monitor patients who are receiving levodopa for changes in neurological status during treatment with Sephience.
Adverse Reactions
Most common adverse reactions with Sephience (≥2% and > placebo) were diarrhea, headache, abdominal pain, hypophenylalaninemia, feces discoloration, and oropharyngeal pain.
Drug Interactions
Avoid concomitant use of drugs known to inhibit folate synthesis dihydrofolate reductase (DHFR) (e.g., trimethoprim, methotrexate, trimetrexate, pemetrexed, pralatrexate, raltitrexed, and piritrexim) while taking Sephience. Concomitant administration of such drugs may reduce sepiapterin metabolism to BH4. If concomitant use is not avoidable, monitor blood Phe levels.
Sephience and PDE-5 inhibitors (e.g., sildenafil, vardenafil, or tadalafil) induce vasorelaxation and may reduce blood pressure. Monitor for signs and symptoms of hypotension.
For medical information, product complaints, or to report an adverse event, please call 1–866–562–4620 or email at usmedinfo@ptcbio.com.
https://en.wikipedia.org/wiki/Sepiapterin