Foamix Pharmaceuticals Ltd. (Nasdaq: FOMX) (“Foamix” or the “Company”), a specialty pharmaceutical company, announced the U.S. Food and Drug Administration (FDA) approval of its novel Amzeeq (minocycline) topical foam, 4%. Amzeeq, formerly known as FMX101, is indicated for the treatment of inflammatory lesions of non-nodular moderate to severe acne vulgaris in adults and pediatric patients 9 years of age and older and is the first topical minocycline to be approved by the FDA for any condition.
“The FDA approval of Amzeeq is a milestone moment in dermatology and the most significant advancement with minocycline in almost 50 years,” said David Domzalski, Chief Executive Officer of Foamix. “We are proud that our proprietary technology platform has led to this new treatment option, which we believe can help address unmet treatment needs for moderate to severe acne patients. We are looking forward to bringing Amzeeq to market in January 2020, and to our Company’s first commercial launch.”
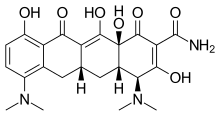
Minocycline is a broad-spectrum antibiotic known for its efficacy in treating moderate to severe acne, but its use is limited in some patients due to systemic side effects when taken orally. Until now, minocycline has not been available as a topical treatment due to its instability in traditional topical formulations. In Amzeeq, Foamix has leveraged its proprietary Molecule Stabilizing Technology (MST™) platform to effectively deliver minocycline in a foam-based vehicle.
“Our innovative MST™ technology allowed us to develop a topical formulation of minocycline in a convenient, once-daily treatment regimen that maintains the stability of the active ingredient while delivering it into the skin,” said Iain Stuart, Ph.D, Chief Scientific Officer of Foamix. “The approval of Amzeeq represents a significant step toward our goal of enhancing the standard of care for the millions of acne sufferers in the U.S. who deserve alternatives in treatment.”
“The approval of Amzeeq is exciting news that provides a much-needed option in the treatment of moderate to severe acne,” said Linda Stein Gold, M.D., Director of Dermatology Clinical Research and Division Head of Dermatology at Henry Ford Health System in Detroit, Michigan. “Minocycline has been a trusted staple in acne treatment for decades, but has only been available in oral or systemic formulations. With the approval of Amzeeq, I can now offer my patients a new, effective topical treatment option with a favorable tolerability profile.”
The FDA approval of Amzeeq is supported by data from three Phase 3 clinical trials in 2,418 patients of 9 years of age or older, making it one of the largest clinical programs for acne to date. In each 12-week, multicenter, randomized, double-blind, vehicle-controlled study, subjects with moderate to severe acne vulgaris were treated once-daily with Amzeeq or vehicle. No other topical or systemic acne medication was permitted to be used by subjects during the study period. The studies each found statistically significant disease improvement with Amzeeq versus vehicle for the co-primary endpoint of absolute reduction of inflammatory lesions, while studies 2 and 3 demonstrated a statistically significant improvement in IGA treatment success. IGA treatment success was defined as a score of 0 (“clear”) or 1 (“almost clear”) and at least a two-point decrease from baseline. Amzeeq was well-tolerated and no treatment-related serious adverse events were reported. The most common adverse reaction was headache, which was reported in 3% of subjects treated with Amzeeq versus 2% of subjects treated with vehicle.
https://en.wikipedia.org/wiki/Minocycline
