Allergan plc (NYSE: AGN), a leading global pharmaceutical company, announced the approval of Byvalson (nebivolol and valsartan) 5 mg/ 80 mg tablets, by the U.S. Food and Drug Administration (FDA) for the treatment of hypertension to lower blood pressure. Byvalson is the first and only fixed-dose combination (FDC) of a beta blocker (BB) and angiotensin II receptor blocker (ARB) available in the U.S.
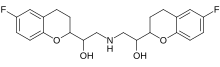 nebivolol
nebivolol 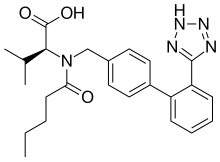 Valsartan
Valsartan
"Achieving blood pressure control is critical to reducing the risk of serious and life-threatening cardiovascular events. There remains a need for new therapies, as observed by the nearly half of patients in the U.S. who remain uncontrolled," said David Nicholson, Chief R&D Officer at Allergan. "We are pleased with the FDA approval of Byvalson, which will provide physicians a new fixed dose combination therapy treatment option for patients affected by hypertension."
Hypertension represents a significant public health problem with high prevalence in the U.S. Hypertension often has no warning signs or symptoms and has been associated with serious cardiovascular (CV) risks, such as stroke, heart failure, and myocardial infarction. Lowering blood pressure reduces the risk of fatal and nonfatal cardiovascular events, primarily strokes and myocardial infarctions. These benefits have been seen in controlled trials of antihypertensive drugs from a wide variety of pharmacologic classes, including the beta-blocker class to which nebivolol principally belongs and the ARB class to which valsartan principally belongs. There are no controlled trials demonstrating risk reduction with Byvalson.
"The majority of patients with hypertension require two or more medications to achieve their blood pressure goals," said William B White, MD Professor of Medicine and Chief of the Division of Hypertension and Clinical Pharmacology, Calhoun Cardiology Center at UConn Health in Farmington and immediate past president of the American Society of Hypertension. "Nebivolol and valsartan are used widely in the management of hypertension and are effective drugs. The new fixed-dose combination Byvalson, that includes these 2 therapies, offers reduction of blood pressure through multiple mechanisms of action."
The FDA approval of Byvalson was based on a Phase 3, double-blind, placebo-controlled, dose-escalating, 8-week efficacy and safety study, published in The Lancet, which randomized approximately 4,100 patients with Stage 1 or 2 hypertension. In this pivotal efficacy and safety study, treatment with the FDC of nebivolol and valsartan for 4 weeks demonstrated statistically significant reductions from baseline in diastolic and systolic blood pressure versus either nebivolol alone or valsartan alone. The overall rate of adverse events was similar across treatment groups and placebo during this 4 week period.
