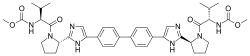
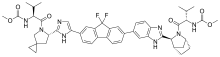
This triple combination treatment was generally safe and effective, even in patients who had been unsuccessfully treated with direct acting antivirals (DAAs, medicines which have been used to treat and cure almost all patients with HCV). The study showed that 99% of patients with HCV genotype 1, 2, 3, 4 and 6 who had previously received treatment, achieved SVR 12 weeks after treatment using this triple combination.
Hepatitis C is a virus carried via the blood, which infects and damages the liver.1 HCV infects liver cells, resulting in inflammation and fibrosis.1 In chronic HCV cases, such symptoms may continue to increase and result in liver cirrhosis, scarring of the liver.1 Despite the high overall SVR rate achieved with currently approved DAA therapies, approximately 5% of patients treated with DAAs will not be cured.2 According to the study authors, for this small proportion of patients who are not cured, retreatment options are significantly limited.
"Our study demonstrates that for HCV patients whose prior treatment has failed with the use of DAAs, this triple combination provides a high rate of sustained virologic response across HCV genotypes," said Dr Eric Lawitz, Clinical Professor of Medicine at the University of Texas Health Science Center, San Antonio and lead author of the study. "Furthermore, the study indicates that the treatment combination is generally safe and well tolerated by patients, providing a promising alternative for HCV sufferers who have limited re-treatment options."
Two global, open-label Phase 2 studies were conducted among chronic HCV-infected patients that had failed prior HCV treatment. Genotype 1 HCV-infected patients enrolled in the study had previously been treated with an NS5A-inhibitor or multiple classes of DAAs, and genotype 2-6 HCV-infected patients had previously been treated with pegylated-interferon (Peg-IFN) plus ribavarin and/or any DAA. All patients received the triple combination of sofosbuvir/velpatasvir plus GS-9857 for 12 weeks. Frequently reported adverse events (AEs) were headache, fatigue, diarrhea and nausea; most were mild or moderate in severity.
"Having offered promising results, this three drug combination is being further evaluated in Phase 3 trials as a single tablet regimen in DAA-experienced patients," said Professor Laurent Castera, EASL Secretary General.
Additional study results
A total of 128 patients were treated: 75% were male, 82% were white, 73% with non-CC IL28B genotypes, and 48% with cirrhosis. 49% of patients had genotype 1, 16% had genotype 2, 27% had genotype 3, 5% had genotype 4 and 2% had genotype 6.
Overall, 27% of patients had been previously treated with NS5A-inhibitors, 52% had been previously treated with other DAAs, and 21% had failed interferon-based treatment without a DAA. Baseline resistance-associated variants (RAVs) were detected in 60% of patients (20% with RAVs to NS5A-inhibitors and 23% with RAVs to multiple classes).
The most commonly reported AEs (>10%) were headache, fatigue, diarrhea, and nausea; most were mild or moderate in severity. One patient (<1%) experienced a serious AE of gastroenteritis (<1%) and 1 patient discontinued treatment early due to an AE of gastritis at Week 9 and achieved SVR12; both AEs were not considered related to the study drugs. No clinically significant laboratory abnormalities were observed
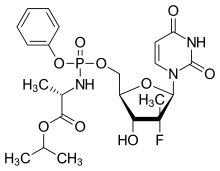


 Sofosbuvir
Sofosbuvir