KemPharm, Inc. a clinical-stage specialty pharmaceutical company engaged in the discovery and development of proprietary prodrugs, announced that the U.S. Food and Drug Administration (FDA) has issued a Complete Response Letter (CRL) regarding the New Drug Application (NDA) for Apadaz™ (benzhydrocodone and acetaminophen), KemPharm’s investigational abuse-deterrent product candidate for the short-term management of acute pain.
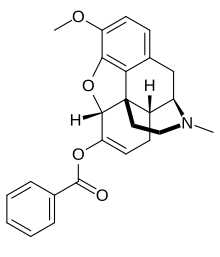 benzhydrocodone
benzhydrocodone 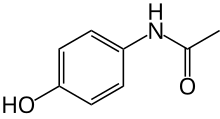 acetaminophen
acetaminophen
The FDA issues CRLs to indicate that the Agency considers the review cycle for an application is complete and that the application is not ready for approval in its present form. Included in the CRL is guidance that describes all specific deficiencies that the FDA has identified in the application. When possible, the FDA recommends actions that the applicant may take to place the application in condition for approval.
“After last week’s amendment request, a Complete Response Letter from the FDA was received for the Apadaz NDA,” said Travis C. Mickle, Ph.D., President and CEO of KemPharm. “We are currently evaluating the points raised in the CRL and intend to request an End of Review meeting with the Agency to determine the pathway forward for Apadaz.”

No comments:
Post a Comment